Advancing Immunotherapy Research: Syngeneic Tumor Models for Preclinical Success
Empower Your Research with TD2’s Syngeneic Tumor Models
TD2 executes data driven in vivo studies using well characterized syngeneic tumor models. These models are critical for evaluating immune checkpoint inhibitor efficacy within a fully functional immune system. Performing preclinical in vivo studies using meticulously characterized syngeneic models ensures relevance and reliability in preclinical oncology research. By utilizing TD2 for preclinical immunotherapy studies, researchers can obtain accurate insights into tumor-immune interactions, facilitating the development of novel immunotherapeutics.

Features and Benefits:
- Fully Characterized: TD2 executes preclinical in vivo studies in syngeneic tumor models that are fully characterized for gene expression, TIL baseline populations, and response to common immune checkpoint inhibitors.
- Diverse Tumor Types: A wide selection of well characterized syngeneic tumor models enables research across a wide range of cancer types, enhancing the applicability of findings. TD2 has experience performing successful in vivo studies using difficult to develop models such as TC-1 and commonly used models like MC38.
- Predictive Value for Clinical Efficacy: Performing preclinical in vivo studies using well characterized syngeneic tumor models enables a researcher to move into the clinic with confidence. Giving them access to insights to potential clinical outcomes of immunotherapeutic agents.
- Enhanced Immunotherapy Evaluation: TD2’s use of syngeneic models facilitates detailed analysis of immunotherapy efficacy, offering a comprehensive view of how these treatments interact with the immune system to target cancer cells. This feature is especially beneficial for developing and testing new immune checkpoint inhibitors, providing a robust platform to optimize therapeutic strategies before clinical trials.
Learn more about our syngeneic services.
Contact our experts to help advance your immunotherapy candidate with our trusted preclinical research services.
Syngeneic Tumor Models Available for Studies at TD2
| Cancer Type | Syngeneic Line | Expression Data Available | ||||
|---|---|---|---|---|---|---|
| PD-1 | PD-L1 | PD-L2 | CTLA4 | Other | ||
| Bladder | MB-49 MBT-2 |
|||||
| Brain | GL261 | |||||
| Breast | 4T1 4T1-Luc EMT-6 EO771 |
|||||
| Colon | Colon 26-Luc colon 26 CT-26 MC-38 |
TIM-3, OX40 CD40 |
||||
| Hematologic | EL-4 (Lymphoma) MPC-11 (Myeloma) |
Growth Kinetics Growth Kinetics |
||||
| Hepatoma | Hepa 1-6 | |||||
| Lung | KLN205 LL/2 LL/2-Luc TC-1 (HPV-Associated) |
Growth Kinetics | ||||
| Ovarian | ID8-Luc | Growth Kinetics | ||||
| Mastocytoma | P815 | Growth Kinetics | ||||
| Pancreas | Pan02 Pan02 (Orthotopic) |
TIM-3, GITR | ||||
| Renal | RENCA | |||||
| Sarcoma | K7M2 (IV) | Growth Kinetics | ||||
| Skin | B16-F1 B16-F10 B16-BL6 |
|||||
Figure 1. Growth curve response to standard of care treatments in orthotopic E0071 in C57BL/6 mice, subcutaneous MC38 in C57BL/6 mice, and subcutaneous CT-26 in balb/c mice in

E0771 Murine Breast Tumor Model

MC38 Murine Colon Tumor Model

CT-26 Murine Colon Tumor Model

IMMUNO-ONCOLOGY
Comprehensive preclinical services for immuno-oncology.
TD2’s extensive knowledge of CAR-T in both solid and hematologic tumors enables rapid evaluation of your CAR-T cell therapy, moving your therapeutic program forward to the clinic.
Large panel of human and murine tumor models
- 8 years of experience in cell therapy
- Expertise includes: CAR-T, NK-CAR, IPS-NKs, Macrophages
Humanized mice
- Human PBMCs or effector cell engraftment
- Sourced CD34+ humanized mice
- Humanized immune checkpoint inhibitor mice
Additional Resources
Preclinical, Posters and Publications
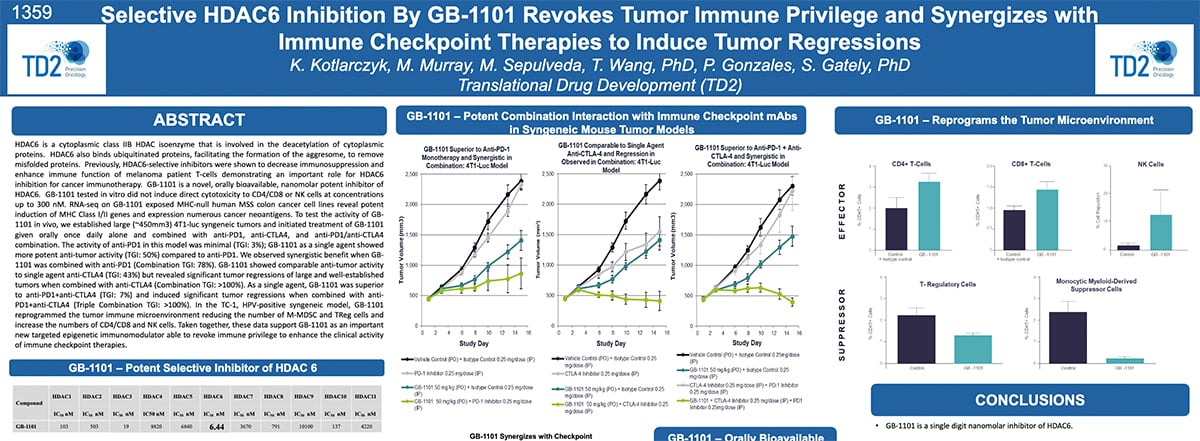
Selective HDAC6 Inhibition By GB-1101 Revokes Tumor Immune Privilege and Synergizes with Immune Checkpoint Therapies to Induce Tumor Regressions
Preclinical, Posters and Publications
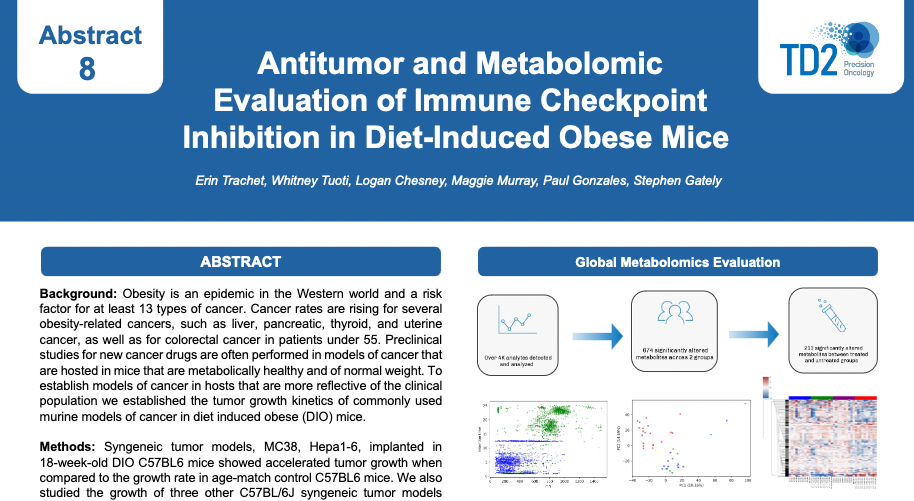
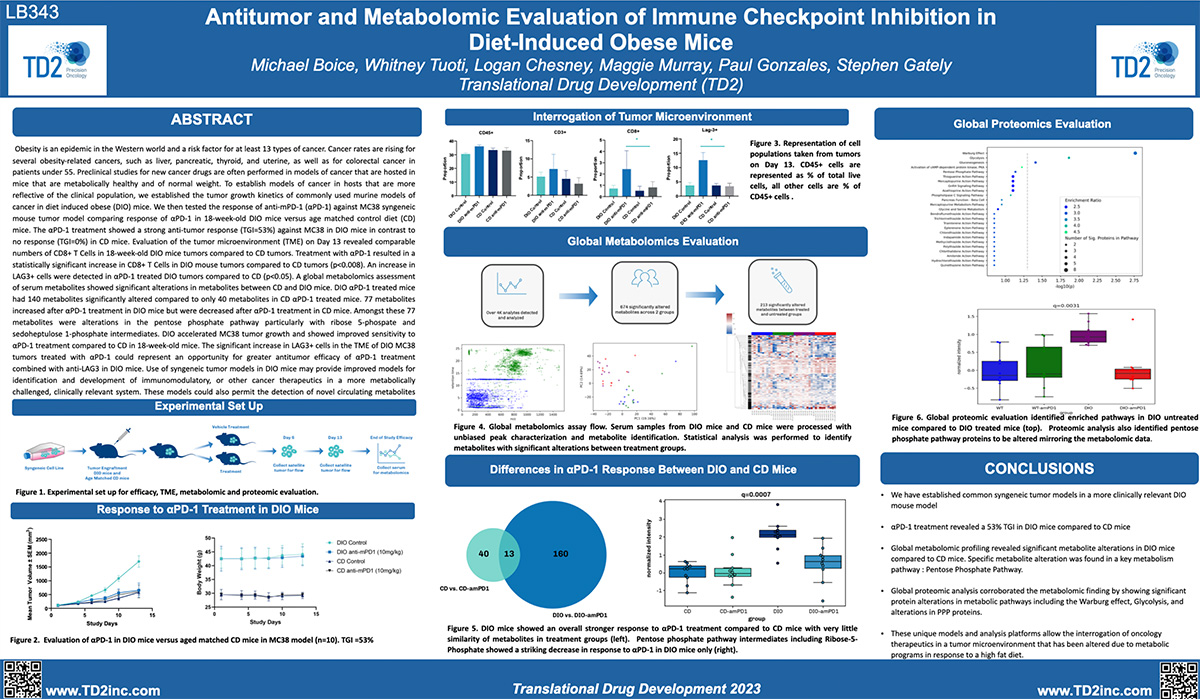
SITC 2023 Poster: Antitumor and metabolomic evaluation of immune checkpoint inhibition in diet-induced obese mice
Preclinical, Posters and Publications
Metabolomics Sample Data Analysis Report
Preclinical, Posters and Publications
Antitumor and metabolomic evaluation of immune checkpoint inhibition in diet-induced obese mice
Videos
TD2 Immuno-Oncology
GET STARTED
Work with a team who believes in your research as much as you do.
Are you ready to start your preclinical Adoptive Cell Transfer Therapy studies? Partner with a collaborative oncology CRO that believes in your treatment as much as you do. Take the first step today and contact our experts.