
Press Release|May 27, 2026
TD2 Oncology and Panome Bio Partner to Advance Translational Oncology Through Integrated Multi-Omics Capabilities
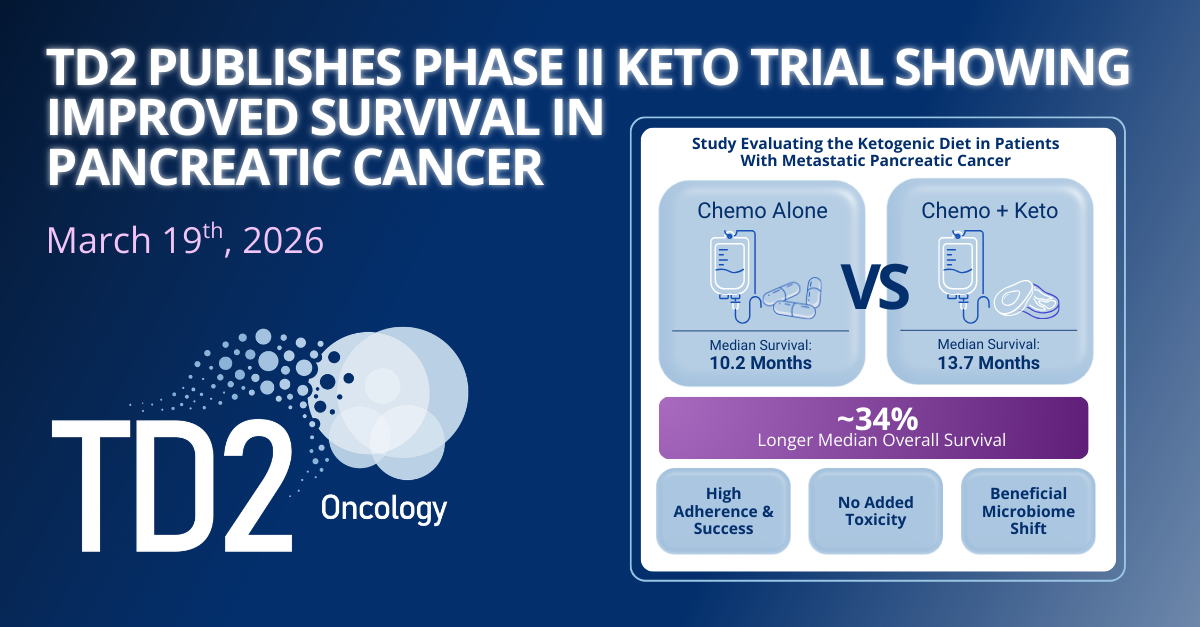
Press Release|March 19, 2026
TD2 Announces Publication of Phase II Ketogenic Diet Trial Showing Improved Survival in Metastatic Pancreatic Cancer

Article|March 26, 2026
How TD2 is Strengthening Site Collaboration Through Data and Shared KPIS

Podcast|December 17, 2025
Steve Gately Joins Bourne Partners for a Deep Dive on the Future of Oncology CROs

News|December 11, 2025
TD2 Oncology Expands Flow Cytometry Expertise to Advance In Vivo CAR T Therapies for Autoimmune and Oncology Development

FIRESIDE CHAT
FIH Clinical Trial Planning for Radiopharmaceuticals: From IND to Patient Dosing
Guiding Oncology Therapies from Early Detection of Response to Approval
Partner with a collaborative oncology CRO
that believes in your research as much as you do.
We Have You Covered Throughout Every Phase
Clinical Trial Services
Preclinical
Regulatory Support
Flow Cytometry
The TD2 Oncology Ecosystem
At TD2, we are revolutionizing oncology research through an integrated ecosystem that brings together preclinical services, advanced flow cytometry, robust regulatory support, an extensive site network, and comprehensive clinical trial services. Our mission is to accelerate the development of innovative cancer therapies.
Learn moreAccelerating Cancer Drug Development
Since 2003, TD2 has made an impact beyond what is expected of a traditional oncology CRO. Not only do we work with hundreds of biotech and pharma companies to rapidly move new therapy discoveries to market through proactive problem solving and strategic collaboration, but we are also drug development innovators. The breadth of our services gives us the unique ability to translate scientific hypotheses generated in a lab setting into patient responses in the clinic. Our results consistently outpace our competition due to a dynamic, flexible and highly specialized team that is laser-focused on oncology.
meet our teamCONTACT US
Work with a team who believes in your research as much as you do.
Are you ready to move your cancer therapy to market? Partner with a collaborative oncology CRO that believes in your treatment as much as you do. Take the first step today and contact our experts.
Latest blog Posts
- How the FDA’s Plausible Mechanism Framework Signals the Future of Precision OncologyClinical|March 13, 2026
- What Makes Radioligand Therapy IND Strategy Fundamentally Different?Regulatory|March 4, 2026
- Receptor Mediated Therapies in the Clinic: From Targeting to ImpactJanuary 7, 2026


