Diet Induced Obesity Model for Cancer Research
A More Clinically Relevant Model to Uncover Your Treatment’s Potential
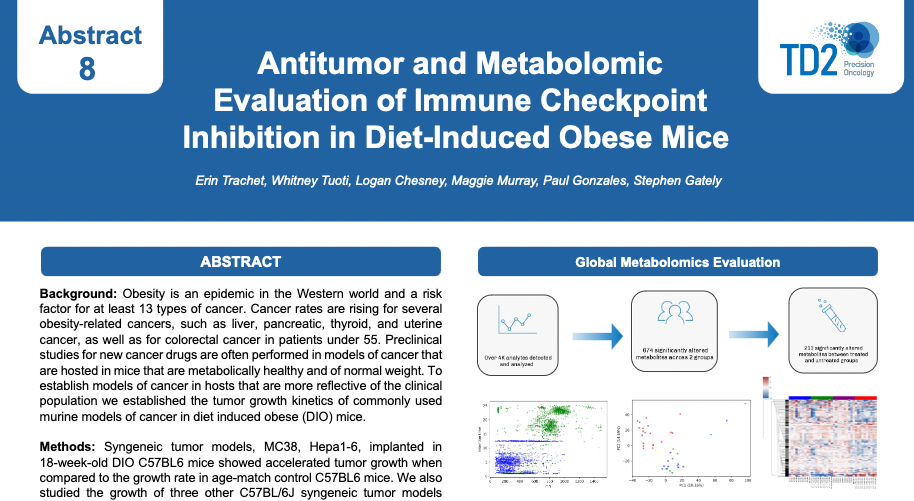
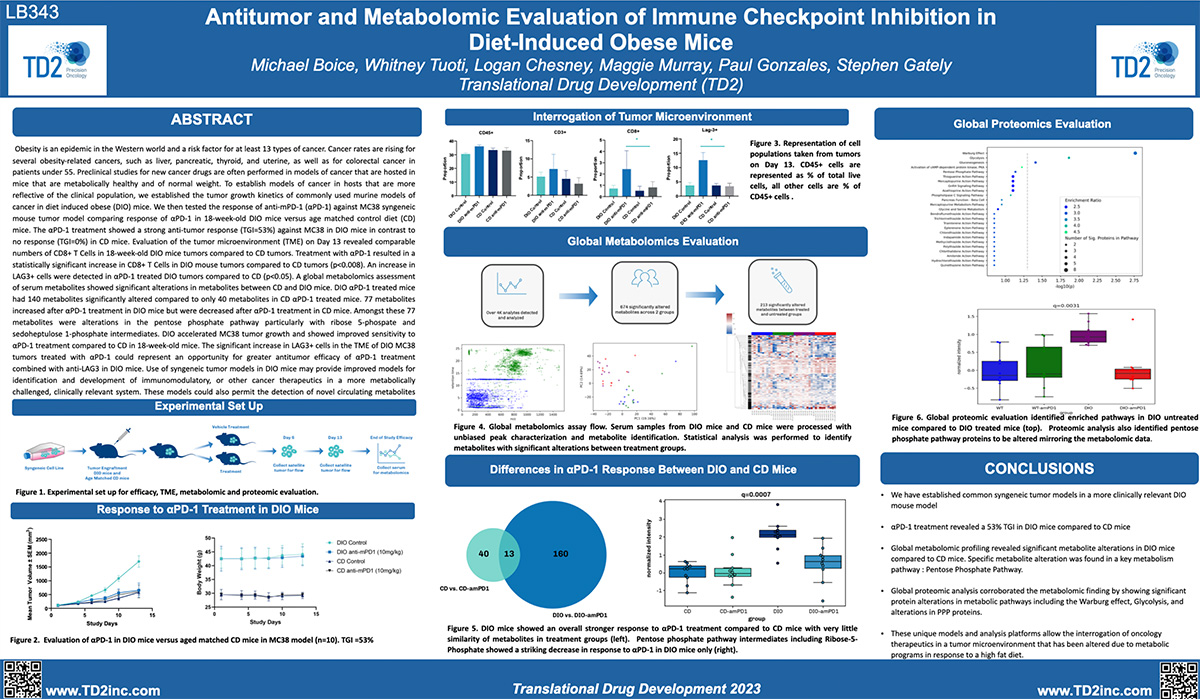
Obesity is a risk factor for at least 13 different forms of cancer. Mechanisms in which obesity and changes in systemic metabolism affect the tumor microenvironment (TME) and impact antitumor immunity are unclear. Current preclinical models use mouse strains which are fed a controlled diet and therefore host mice are not representative of Western human cancer populations. TD2 has established a diet induced obesity tumor model (Figure 1) to screen cancer therapies in a more clinically relevant system.
TD2 integrates the use of syngeneic tumor models with the DIO mouse model to enhance the clinical relevance of preclinical, immune checkpoint inhibitor in vivo studies. This innovative approach allows for the evaluation of cancer therapies within an obese phenotype, which closely mirrors the metabolic and immune conditions present in a significant portion of the cancer patient population. By utilizing syngeneic tumor models in DIO mice, TD2 provides a comprehensive platform that captures the complex interplay between tumor growth, obesity, and the immune system. This platform is particularly important for understanding how obesity alters the tumor microenvironment and affects the efficacy of cancer treatments, including immunotherapies.

Figure 1: Establishment of diet induced obesity mouse tumor model
Benefits of Diet Induced Obesity Mouse Models:
- Execute studies in more clinically relevant models reflective of the significant rates of obesity projected by 2035.
- Understand how obesity induced systemic changes in metabolism effect the tumor microenvironment
- Identify new treatments/mechanism of action that may show increased therapeutic benefit in DIO models
- Quantify metabolite and proteomic changes in response to treatment that could be used as biomarkers of treatment benefit or as opportunity for new targets for drug development

Figure 2: Growth of subcutaneous MC38 model in C57BL6 mice (n=10) and
evaluation of anti-PD1 in DIO mice versus age matched Controlled Diet (CD) mice dosed Q3D x 4
Learn more about our diet induced obesity models.
Contact our experts to help advance your immunotherapy candidate with our trusted preclinical research services.
![]()
Diet Induced Obesity Model Impact on ICI Treatment

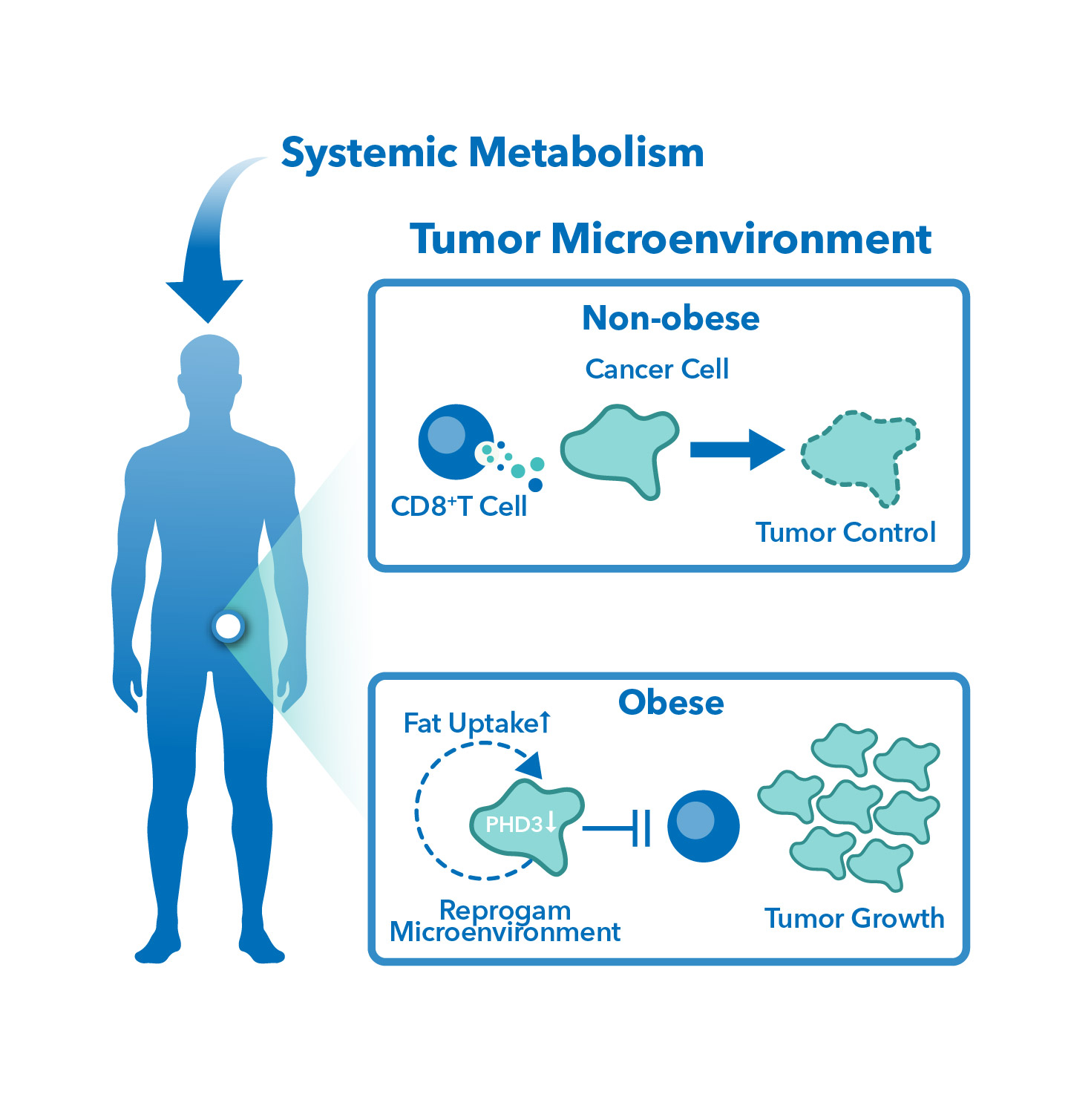
One of the major benefits of using diet-induced obesity (DIO) in mouse models for cancer research is that the diet-induced obesity itself can be manipulated to study how this condition can modulate cancer progression. DIO mouse models can help us further elucidate the mechanisms underlying these cancers. Additionally, DIO mouse models can be used to investigate the bi-directional relationship between obesity and cancer including direct tumor growth and the effects of obesity on cancer therapies.

Significant metabolite alterations between treated
and untreated DIO and CD mice
DIO mouse models offer the potential to study how diet-induced obesity can modulate the effects of different cancer treatments. It is believed that changes in metabolic factors, such as glucose and fatty acid metabolism, as well as other hormones, may influence the efficacy of treatments. DIO mouse models can help to elucidate these changes, aiding the development of targeted therapies for patients.
- DIO mice showed stronger metabolic response to amPD-1
- Very little similarity between treatment groups
- Multiple metabolites showed differential response in DIO vs CD

Comprehensive Preclinical Services
Regardless of the development question, TD2 has the specialized model you need to move your drug forward. Clients have access to humanized models, induction models, as well as specialty surgical and orthotopic models. We specialize in all areas of oncology and work diligently to understand your therapeutics’ mechanism of action. We match that with clinical development strategies for an efficient path forward.
- Non-GLP Safety/Tolerability
- In vitro Pharmacology Assays
- DMPK/ADME
- Orthotopic Tumor Models
- CAR-T and Adoptive Cell Therapy models
- Syngeneic mouse models
- In vivo Optical Imaging
- Metabolomics and Proteomics Analysis
- Humanized Immune Checkpoint Inhibitor Models
- Flow Cytometry
Additional Resources
Preclinical, Posters and Publications
E-Book: Using the Diet Induced Obese Mouse Model for Modern Cancer Research
Preclinical, Posters and Publications
SITC 2023 Poster: Antitumor and metabolomic evaluation of immune checkpoint inhibition in diet-induced obese mice
Preclinical, Posters and Publications
Metabolomics Sample Data Analysis Report
Preclinical, Posters and Publications
Antitumor and metabolomic evaluation of immune checkpoint inhibition in diet-induced obese mice
GET STARTED
TD2 navigates the complexities of drug discovery to guide you down a smarter path for earlier, more complete results.
Contact us today to learn more about our ADME and biomarker offerings and how we can set your treatment up for success from the start.