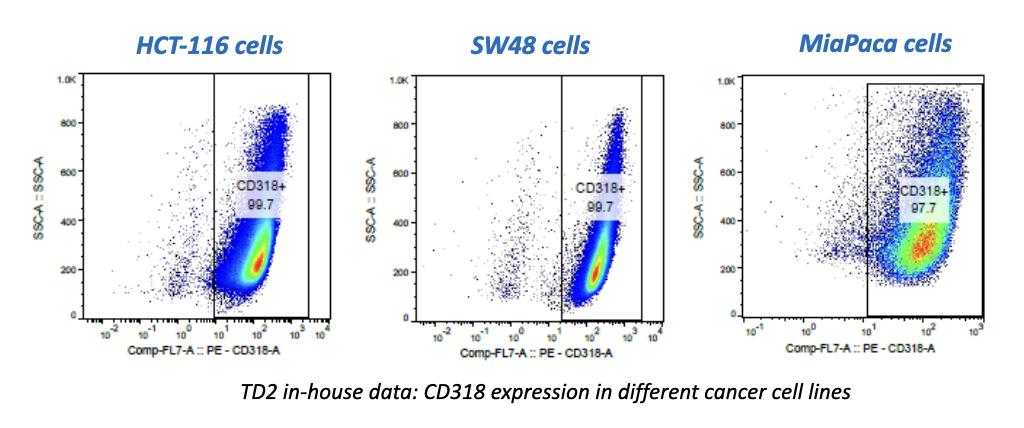
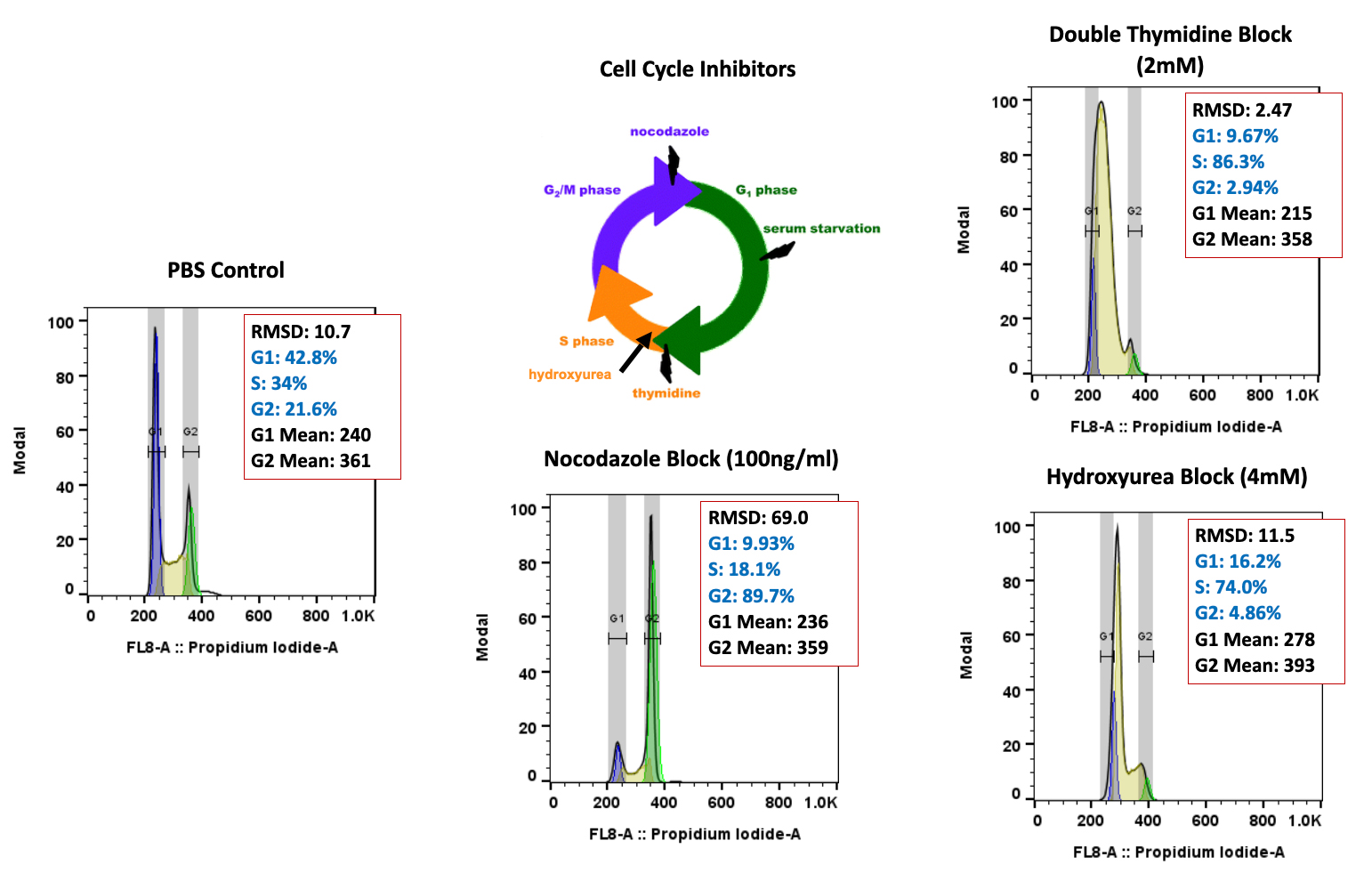
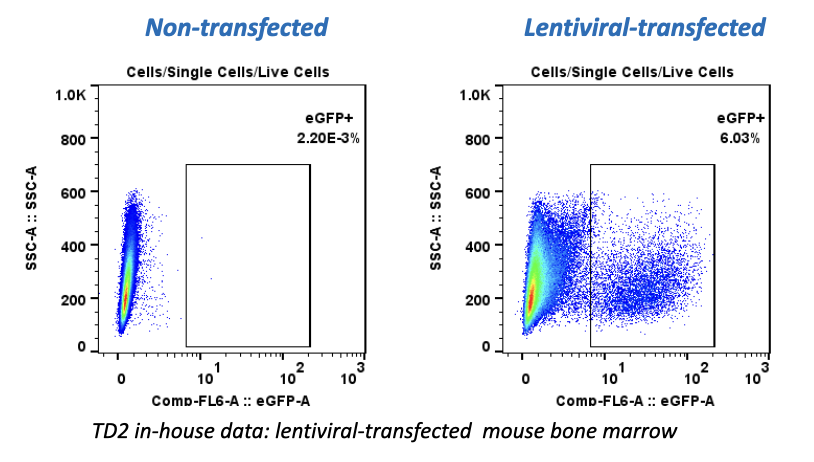
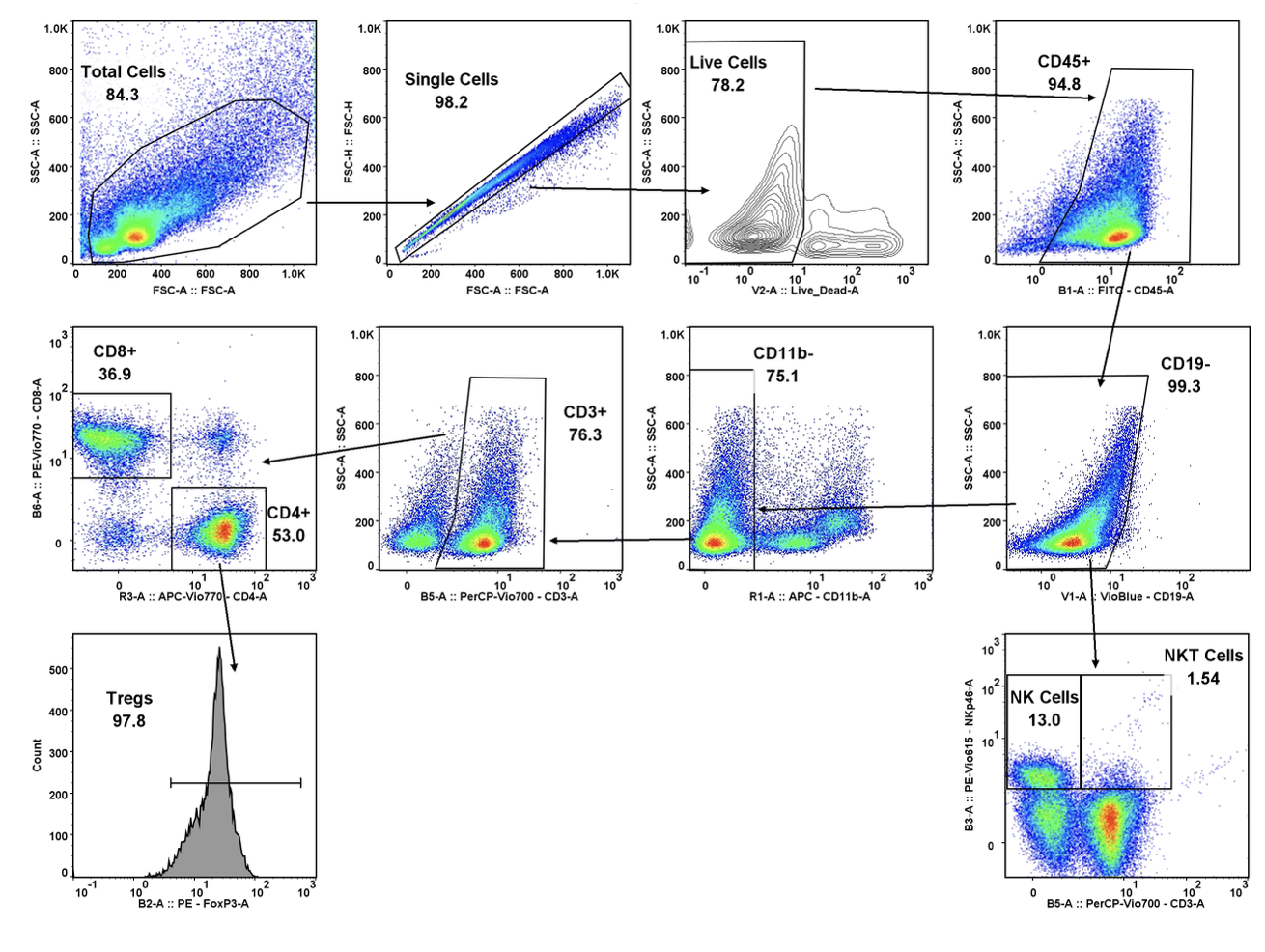
Regulatory Support
Regulatory success. By design.
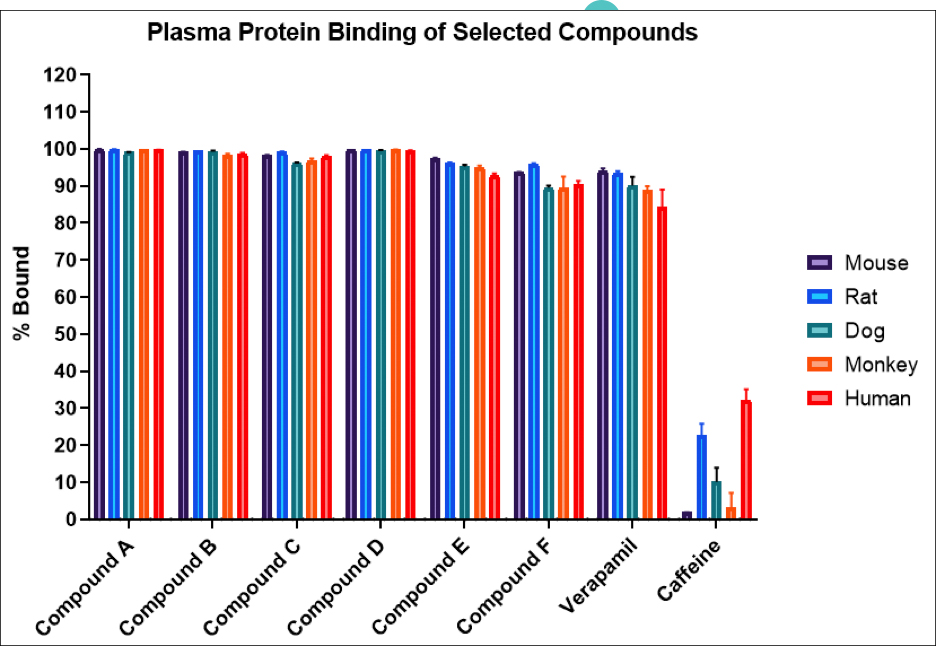
TD2’s Regulatory Affairs Team engages Oncology drug development experts and strategists with proven track records of success. We leverage experienced, oncology-focused medical writers, regulatory experts in CMC, pharmacology, and toxicology, as well as regulatory program managers to provide a customized approach across all areas of drug development.
Translational Regulatory Services
Partner with a trusted, experienced team to guide your preclinical and IND-enabling studies.

Program Management
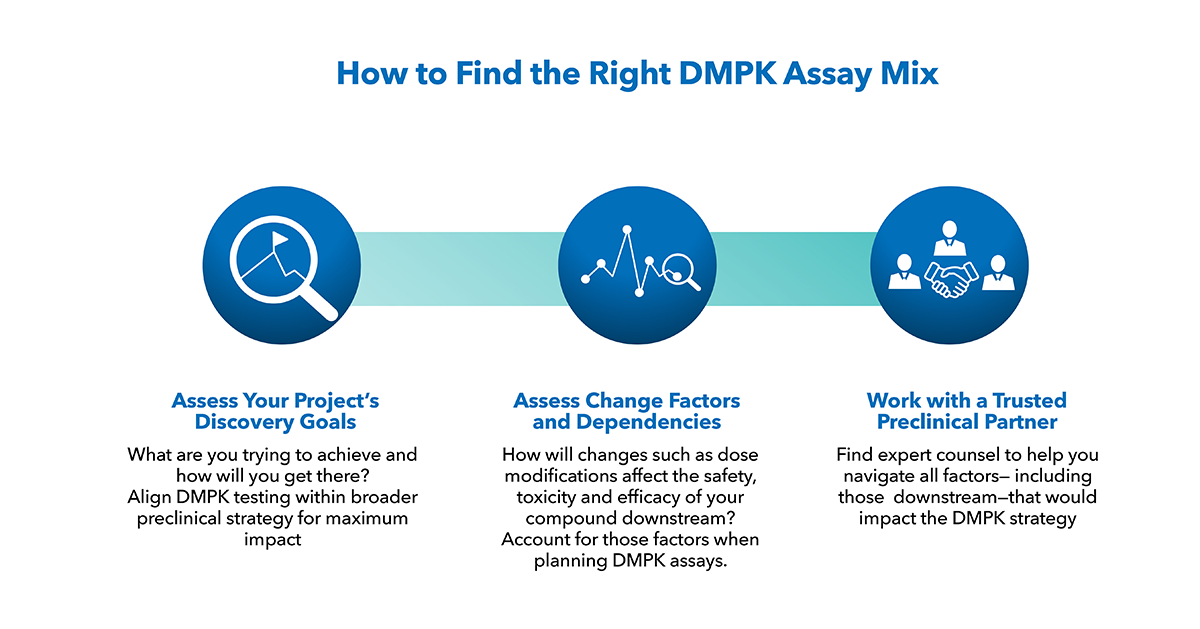
A customized approach to regulatory strategy.
There is no one-size-fits-all approach to regulatory strategy, but with diligent planning and scientific research, we are able to identify unique regulatory opportunities as well as risks that could delay your development program. Chemistry, Manufacturing and Controls (CMC) is a critical path activity that requires careful planning and execution, and we have an expert team to help navigate those challenges. In addition, our Pharmacology and Toxicology experts have decades of experience with small molecules, large molecules and biologics. Close communication keeps your timelines on track moving your therapy forward, faster.