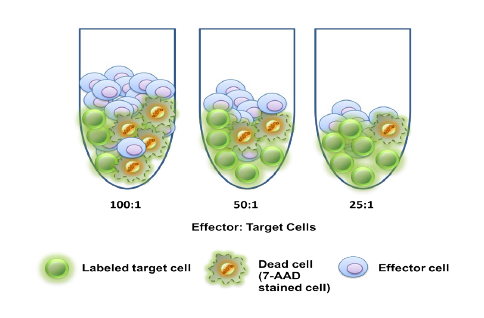
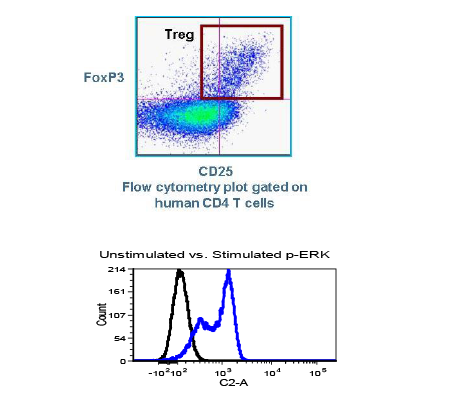
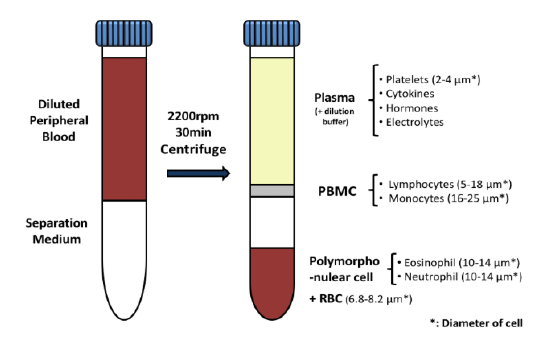
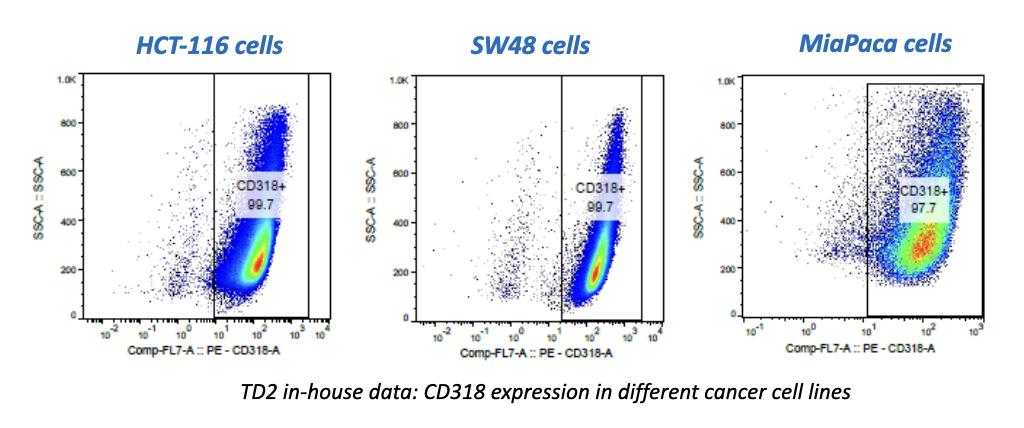
SCOTTSDALE, Ariz. – (June 19, 2013) – Cancer pharmaceutical development company, Translational Drug Development (TD2), and the Translational Research Consortium (TRC) today announced a new partnership to enable rapid testing of anti-cancer drugs combined with radiation therapy. The new TRC-TD2 partnership will allow investigators to test the efficacy of new radiation techniques with potential new drugs, ultimately enhancing treatment options for cancer patients. TRC is the research arm of 21Century Oncology, the nation’s largest radiation oncology provider, and TD2 is an affiliate of the Translational Genomics Research Institute (TGen).
“Nearly two-thirds of all cancer patients will receive radiation therapy during their care,” says Stephen Gately, Ph.D., President and CEO of TD2. “Our partnership will accelerate the identification of medicines that could improve radiation therapy and increase cancer killing capabilities.”
The TRC has installed a new, state-of-the-art research environment with TD2 that enables investigators to mimic the radiation therapy process used when treating patients with cancer. The ARIIEL radiation and immunotherapy laboratory will house the research setup, which provides the most advanced experimental program for testing the efficacy of new radiation techniques and potential new drugs that, together, could enhance the standard of treatments for cancer.
“ARIIEL’s research platform is critical to identify new agents that, when used with radiation therapy, could improve outcomes for patients with cancer,” said Steven Finkelstein, M.D., National Director of the TRC. “This research will be instrumental in shaping future cancer treatment protocols and identifying active combination treatment medicines.”
One of the first applications of this cutting-edge tool is in the area of glioblastoma multiforme, which are aggressive and often deadly brain tumors where front-line treatment is radiation therapy. Because of low cure rates, there is a need to develop new agents and treatments for these tumors. Now, TD2 will be able to simulate the radiation treatment given to patients in a preclinical setting, allowing both TD2 and 21st Century Oncology to identify new treatments for this devastating disease.
By applying cutting-edge preclinical tools, streamlined and efficient regulatory processes, and unique, targeted clinical trial designs and strategies, TD2 facilitates innovative drug development to move new, targeted compounds to patients as quickly as possible. The combination of leading research science and 21st Century Oncology’s clinical expertise will accelerate the development of new agents for cancer patients in Scottsdale and the U.S.