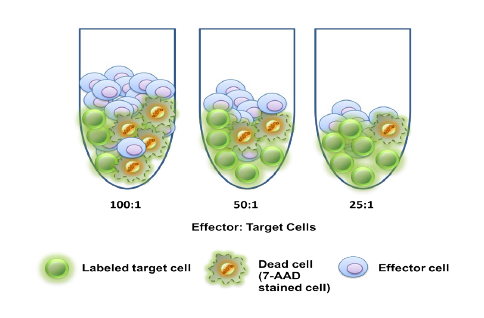
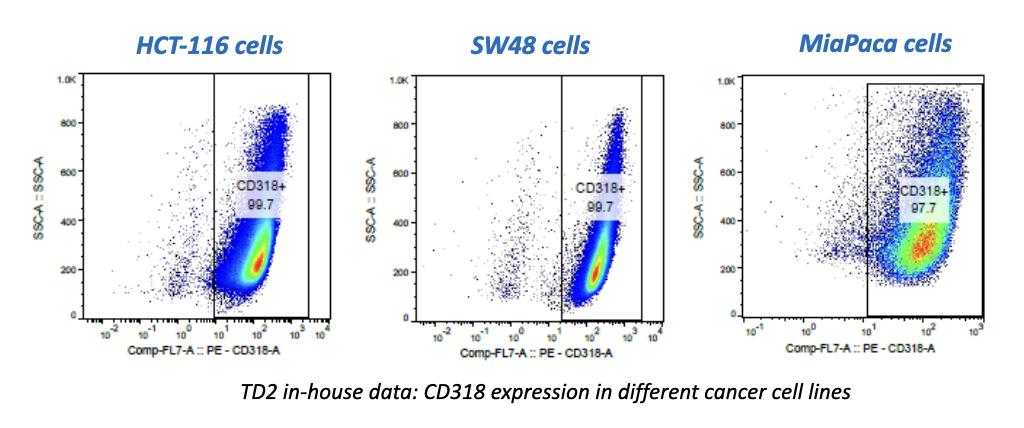
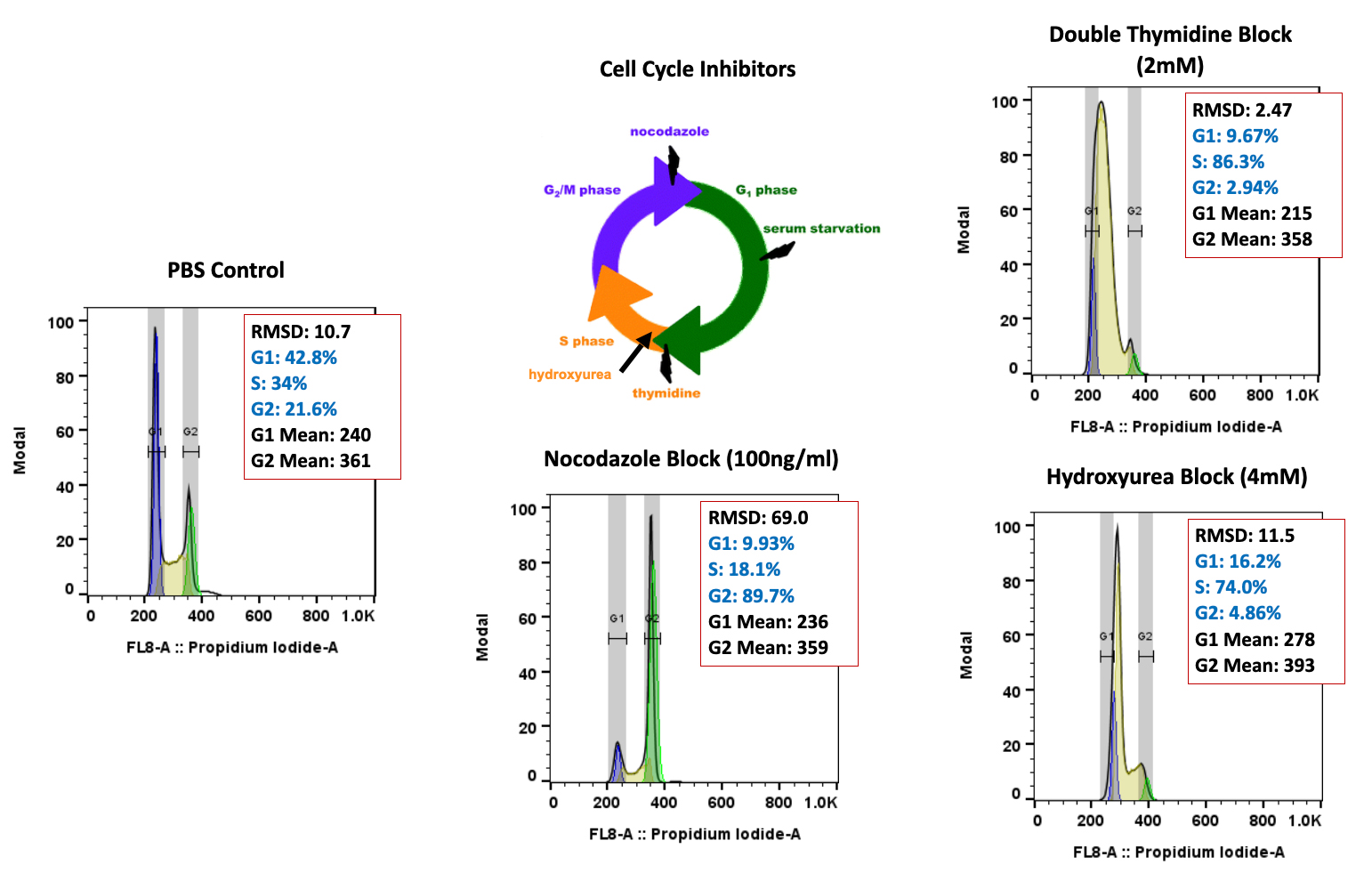
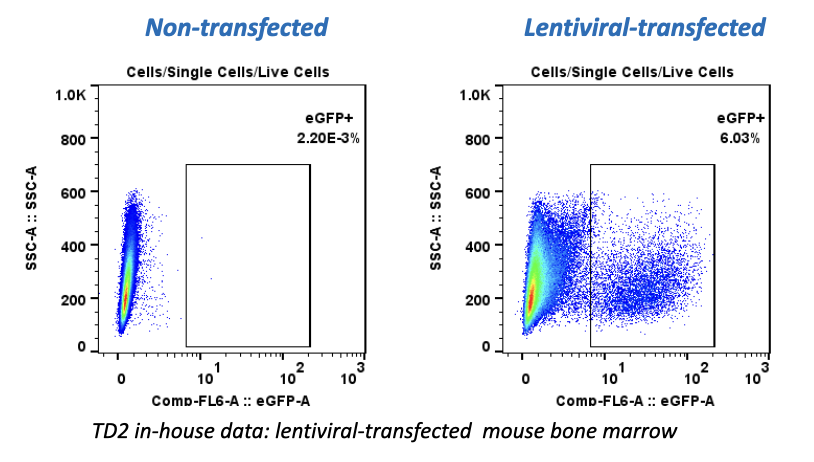
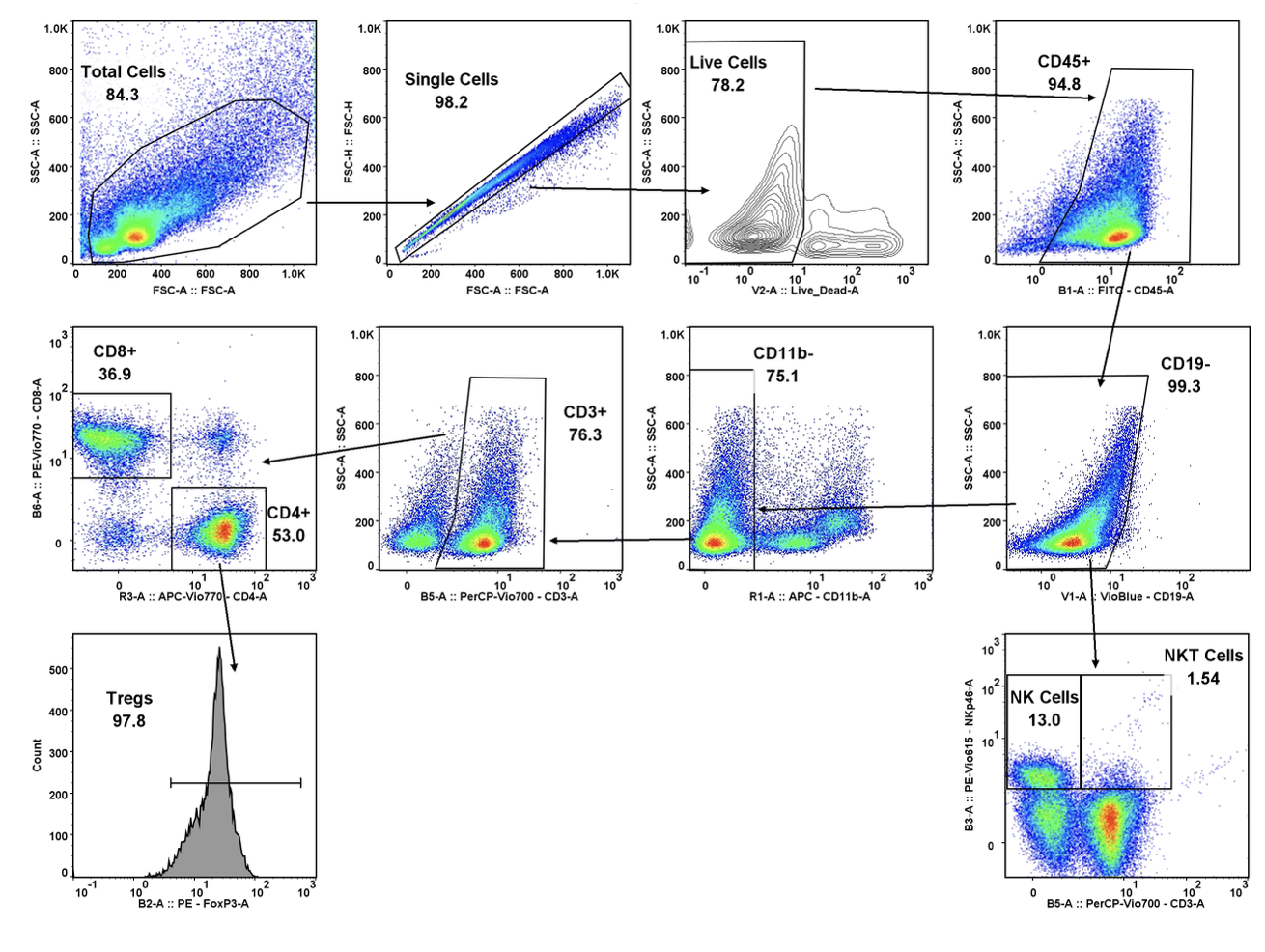
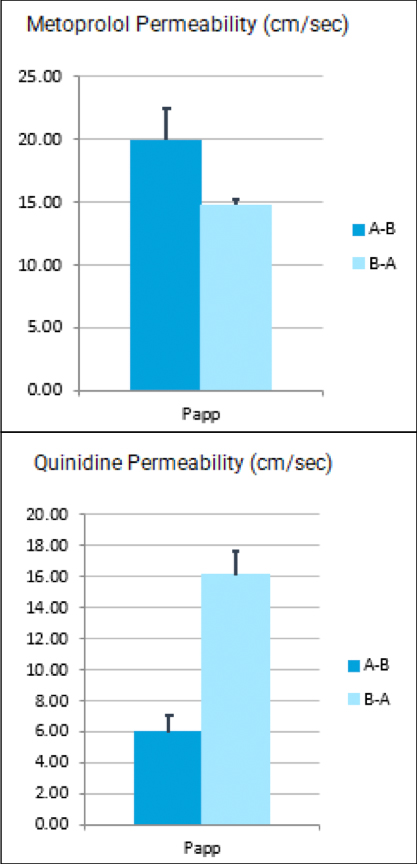
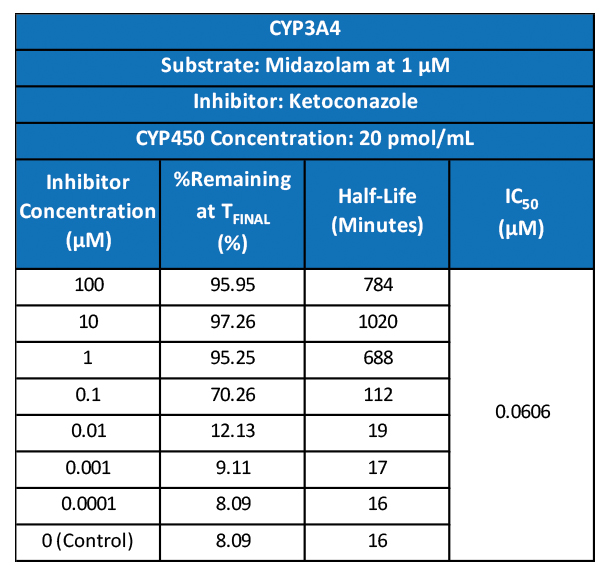
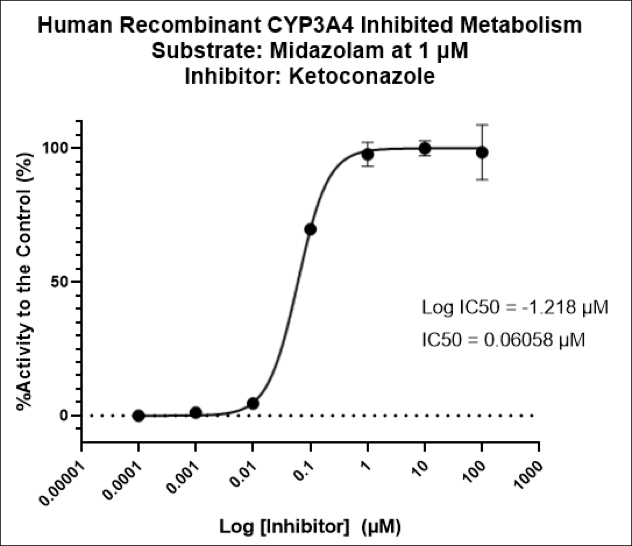
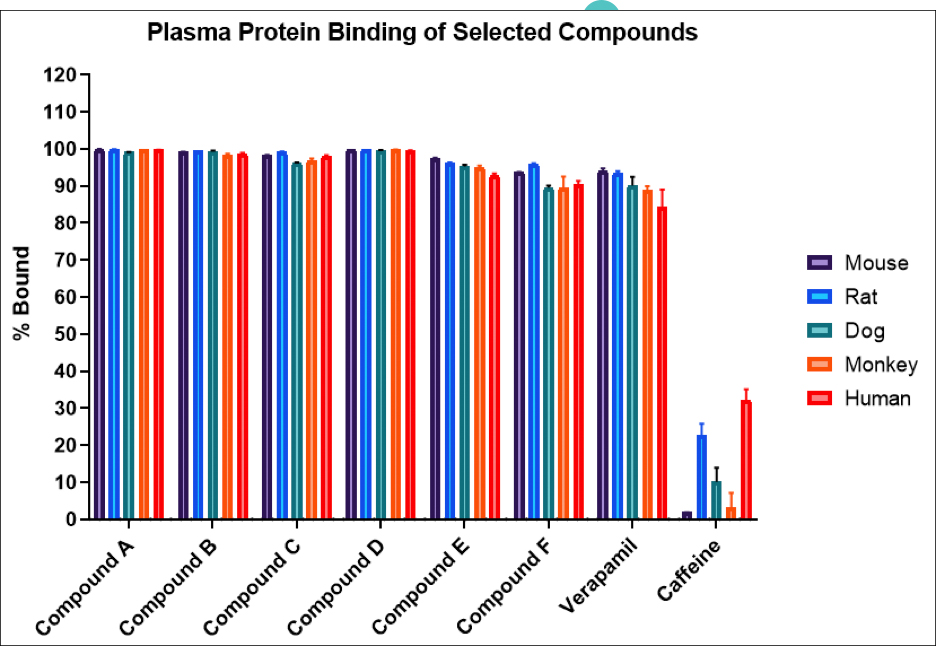
Atossa Genetics Inc. (ATOS) (“Atossa” or the “Company”), a clinical-stage biopharmaceutical company developing novel therapeutics and delivery methods to treat breast cancer and other breast conditions, today announced that it is advancing its intraductal microcatheter immunotherapy program with pre-clinical studies being conducted by Translational Drug Development, LLC (“TD2”). The purpose of the initial study is to develop and validate preclinical methods of using Atossa’s proprietary intraductal microcatheter technology to administer immunotherapy to the site of tumor initiation.
“While there has been recent success treating blood cancers with chimeric antigen receptor therapy (or, CAR-T), there has not been much success using CAR-T to treat solid tumors like breast cancer,” commented Steve Quay, Ph.D., MD, President and CEO of Atossa. “We are, however, encouraged by a recent case study reported June 4, 2018 in Nature Medicine (N. Zacharakis, et al.), in which a patient with metastatic breast cancer who was not responding to chemotherapy had complete tumor regression 22 months after being treated with an intravenous infusion of eighty billion T-cells and interleukin. We believe our proprietary intraductal microcatheter technology may provide a unique and more efficacious and cost-effective treatment method by delivering a significantly smaller number of T-cells directly to the site of the cancer prior to metastasis, rather than through the blood stream, where they are diluted into the entire body. These studies are the first of several steps to develop our intraductal microcatheter technology to treat breast cancer with cell-based immunotherapy, such as CAR-T. These pre-clinical studies will form the basis for the design of human studies, with the ultimate goal of treating breast cancer by administering an immunotherapy with our proprietary microcatheter technology,” added Dr. Quay.
The studies are being conducted for Atossa by Translational Drug Development, LLC, which is an oncology development organization that provides innovative services and is uniquely positioned to support improved and accelerated development of medicines for life-threatening oncology diseases.