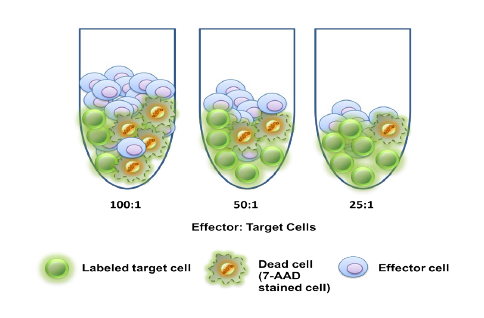
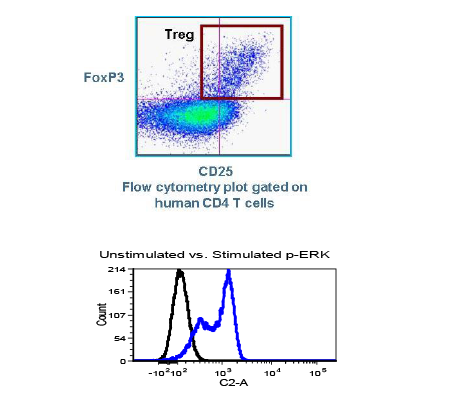
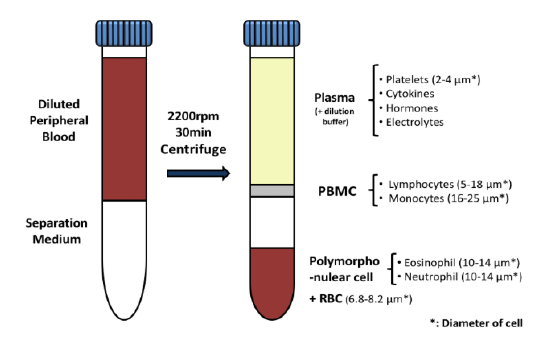
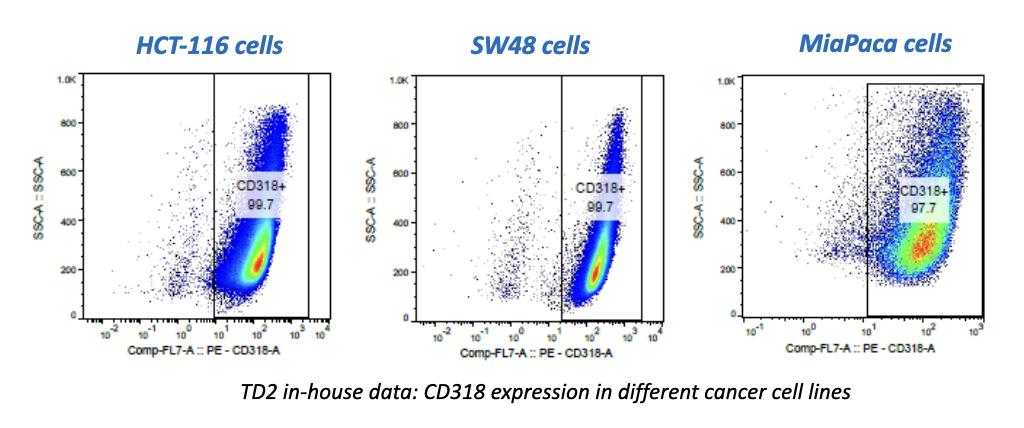
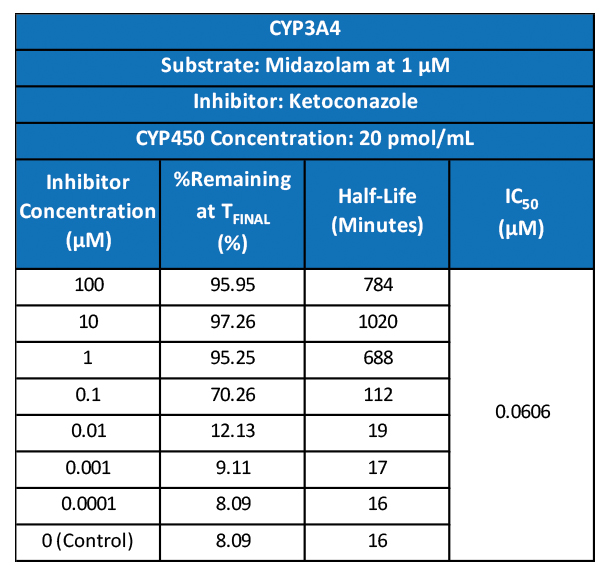
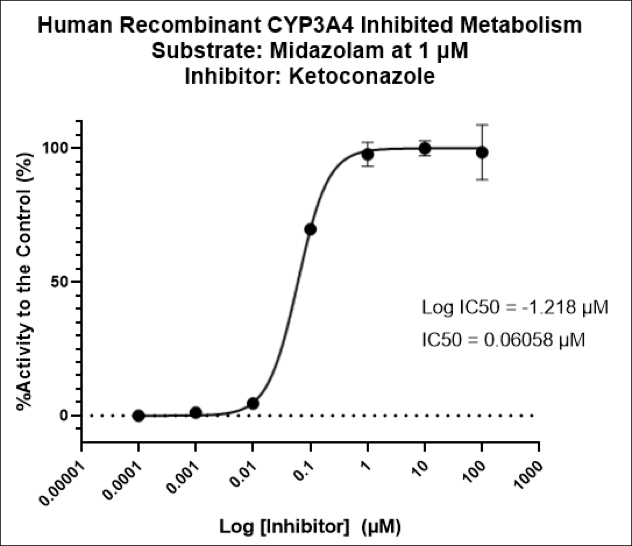
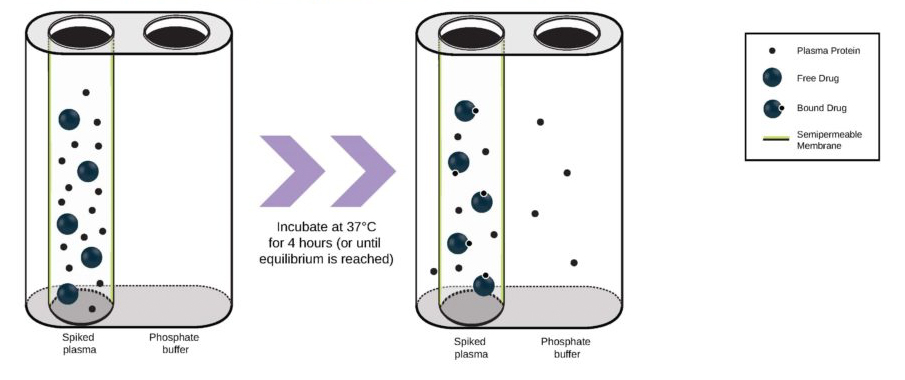
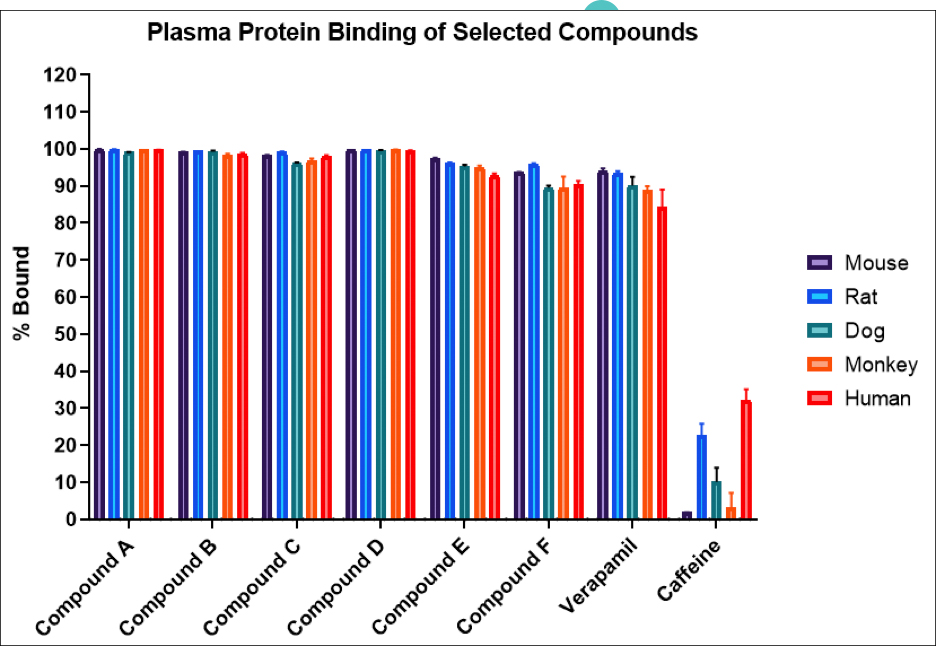
First cancer patient receives dose of super-enhancer inhibitor GZ17-6.02, an advanced cancer drug from Genzada Pharmaceuticals.
Genzada Pharmaceuticals USA Inc.(Genzada), a subsidiary of Ionics Life Sciences Limited, announced today that the first patient enrolled in its Phase 1 clinical trial for the oral therapeutic GZ17-6.02 received the first dose this week at Honor Health Research Institute in Scottsdale, Ariz. Genzada’s GZ17-6.02 will be administered to patients with advanced solid cancers and lymphoma.
The drug is derived in part from the black calla lily (arum palaestinum), a native plant found in several regions of the Middle East. Traditional Middle Eastern medicine has used the lily for more than 1,000 years as an herbal remedy for multiple maladies, including inflammation, viral infection and cancer.
“We’re excited to have reached the clinical stage of development of GZ17-6.02. Through the rigorous preclinical developmental path, we have discovered a unique mechanism of action via super-enhancer inhibition. We’re confident that this agent will be both safe and effective in cancer patients and are thrilled to bring this into the clinic,” said Genzada Chief Operating Officer Cameron West, MD. “Additionally, as GZ17-6.02 is composed of three active pharmaceutical ingredients—originally derived from medicinal plants—we’re hopeful that 6.02 will tackle various malignancies on multiple fronts.”
Genzada chose TD2, a contract research organization (CRO), to manage the operational aspects of the trial, including trial design, project management, data management, monitoring, clinical site management, safety and statistics.
“Reaching the clinical stage is a huge milestone for the companies we work with, as it takes a lot of hard work and preparation to get to this point,” said Stephen Gately, president and CEO of TD2. “This particular drug is quite interesting in its composition, and we’re honored to be part of this trial that could potentially improve the lives of cancer patients.”
With enrollment for the study projected to reach 30-40 patients nationally, Genzada expects continued treatments among trial patients throughout the remainder of 2019. Currently, patients enrolled in this clinical trial will receive the GZ17-6.02 (6.02), an investigational compound,at HonorHealth Research Institute in Scottsdale. In the future, additional trial sites may be included as the study progresses.
“GZ17-6.02 is truly a novel therapy with the potential to transform cancer care. With the support of Genzada and TD2, we are pleased to make this treatment available at HonorHealth Research Institute,” said Frank Tsai, MD, medical oncologist, principalinvestigator at HonorHealth Research Institute.
In November 2018, the project’s Investigational New Drug (IND) application received clearance from the U.S. Food and Drug Administration (FDA) to proceed with the Phase 1 human trial of 6.02 in patients with advanced solid cancers and lymphoma.
The 6.02 compound acts as an inhibitor to super-enhancers, the areas of the genome bound by transcription factors that are uniquely susceptible to repair signal disruption. It previously showed success in many therapeutic settings, including patients with pancreatic cancer and those with head and neck squamous cell carcinoma.