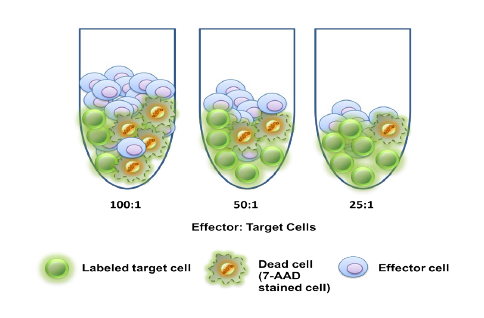
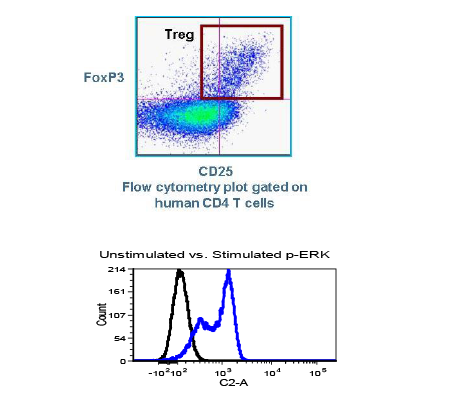
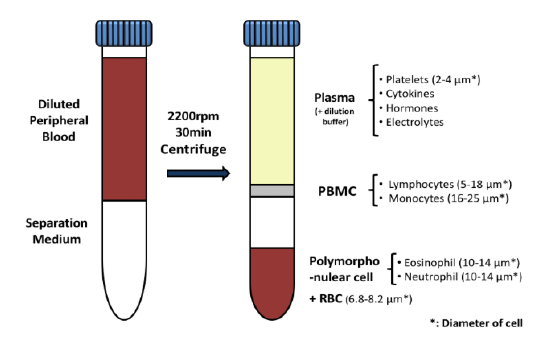
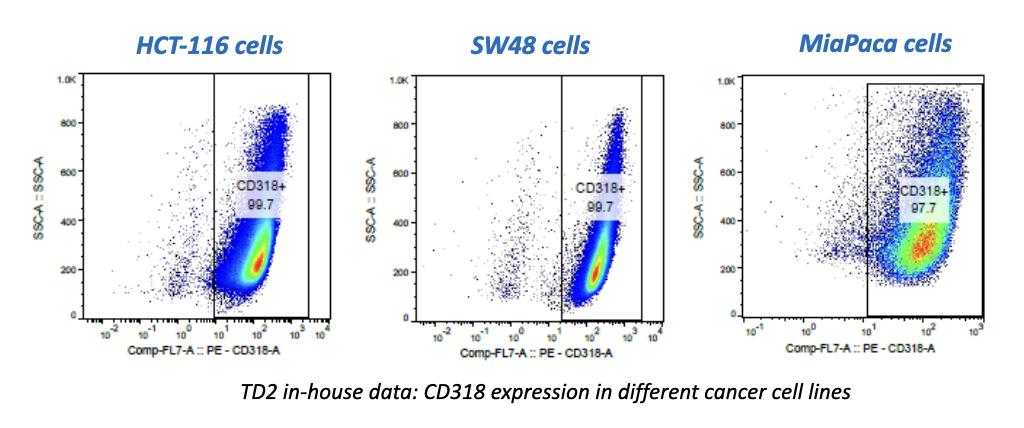
Most people wanting to participate in cancer clinical trials are people suffering advanced cancer. Having depleted treatment options that failed to stop the progression of their cancer, these patients choose to engage in clinical trials offering new and promising anti-cancer treatments. However, many cancer clinical trials involve investigations regarding recently developed methods of detecting or diagnosing cancer using state-of-the-art devices and machines. For these trials, researchers often prefer people with stage 0 or stage I cancer to participate rather than those with advanced cancer. This is because the detection of cancer in its earliest stage is imperative to increasing treatment efficacy and prognoses.
Should I Participate in a Cancer Clinical Trial if I Have Cancer?
Like other cancer treatment options, cancer clinical trials offer both benefits and potential risks, especially regarding the effectiveness of the experimental therapy. Just because a new anti-cancer drug is undergoing trials doesn’t necessarily mean it will slow progression of cancer.
Benefits of being a participant in a cancer clinical trial include having access to a new drug or treatment that could be beneficial and helping researchers learn more about diagnosing and treating different forms of cancer. No health risks are involved with taking experimental cancer drugs in a clinical trial because these drugs have already been tested for several years on animal subjects. Additionally, highly skilled and trained doctors and researchers familiar with the physiological activities induced by cancer cells and tumors constantly monitor the health of all clinical trial participants.
The main risk of advanced cancer patients opting to participate in a cancer clinical trial concerns the efficacy of the new drug. It may work, it may only work slightly or it may not work at all. Participants may also need to visit several physicians, including their primary care doctor providing them with standard treatment. Also, some experimental cancer therapies may help one patient but not other patients.
Who Can Apply to Participate in Cancer Clinical Trials?
All clinical trials implement study plans called protocols describing how the experiment will be performed, whether it is a randomized, double-blind or other structured trial and why each stage of the trial is essential to providing statistically significant results. Protocols also include descriptions of qualifiers for potential study subjects. Eligibility criteria found in most protocols want participants to have a specific stage or type of cancer, have not received or did receive therapies in the past, have special genetic changes in tumors, belong to certain age groups and have a readily available medical history.
People with advanced stage cancer who wish to participate in a cancer clinical trial should discuss this decision with their doctor and family members. Some trials need participants to travel to a facility and remain near the facility during the course of the trial, which may produce a significant lifestyle change for everyone involved.
If you would like more information about advanced cancer subjects in cancer clinical trials, please contact us today.