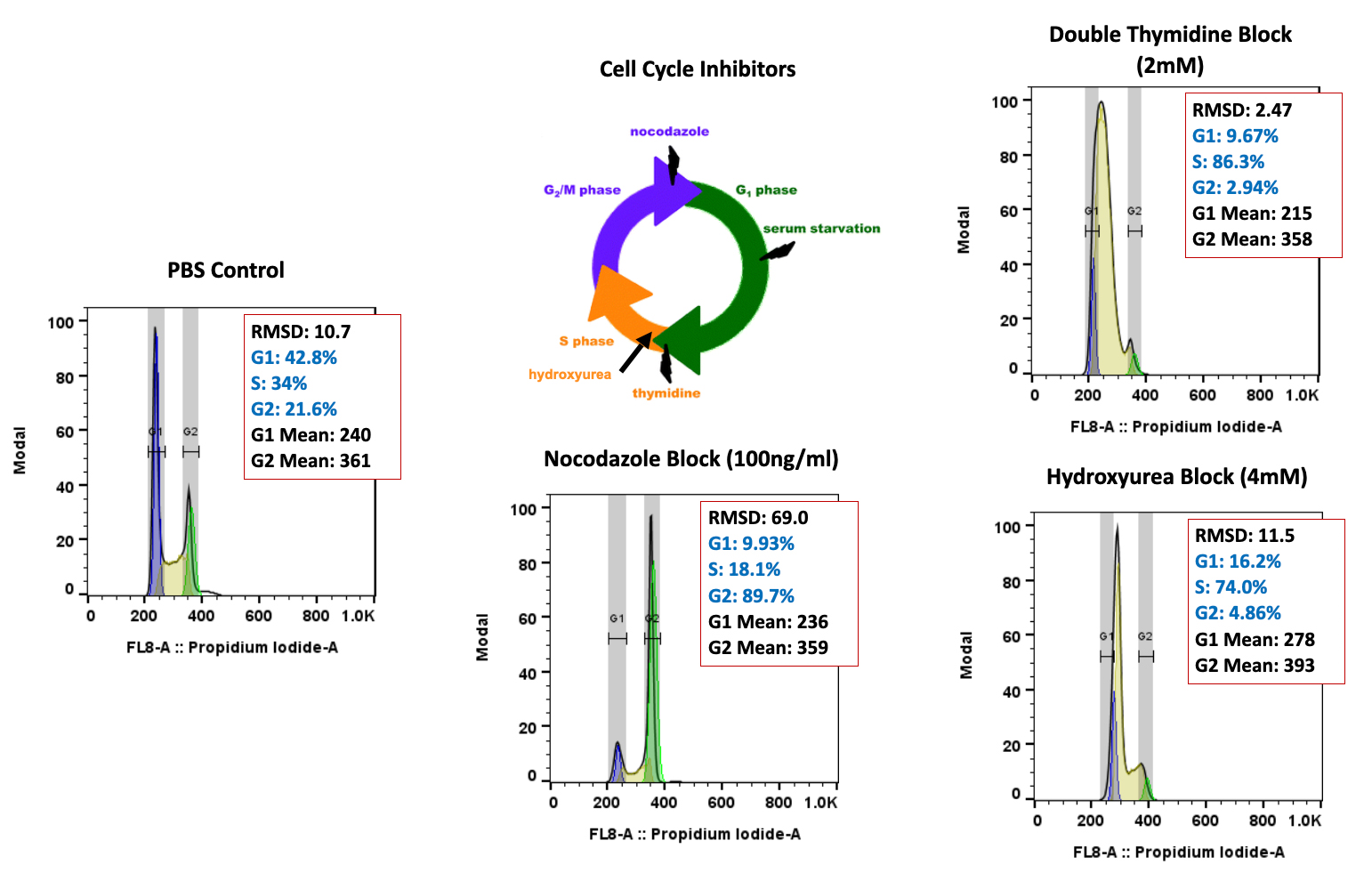
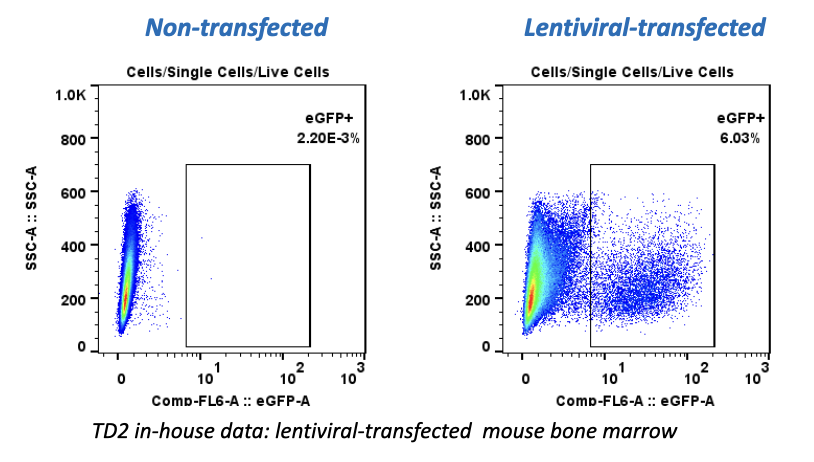
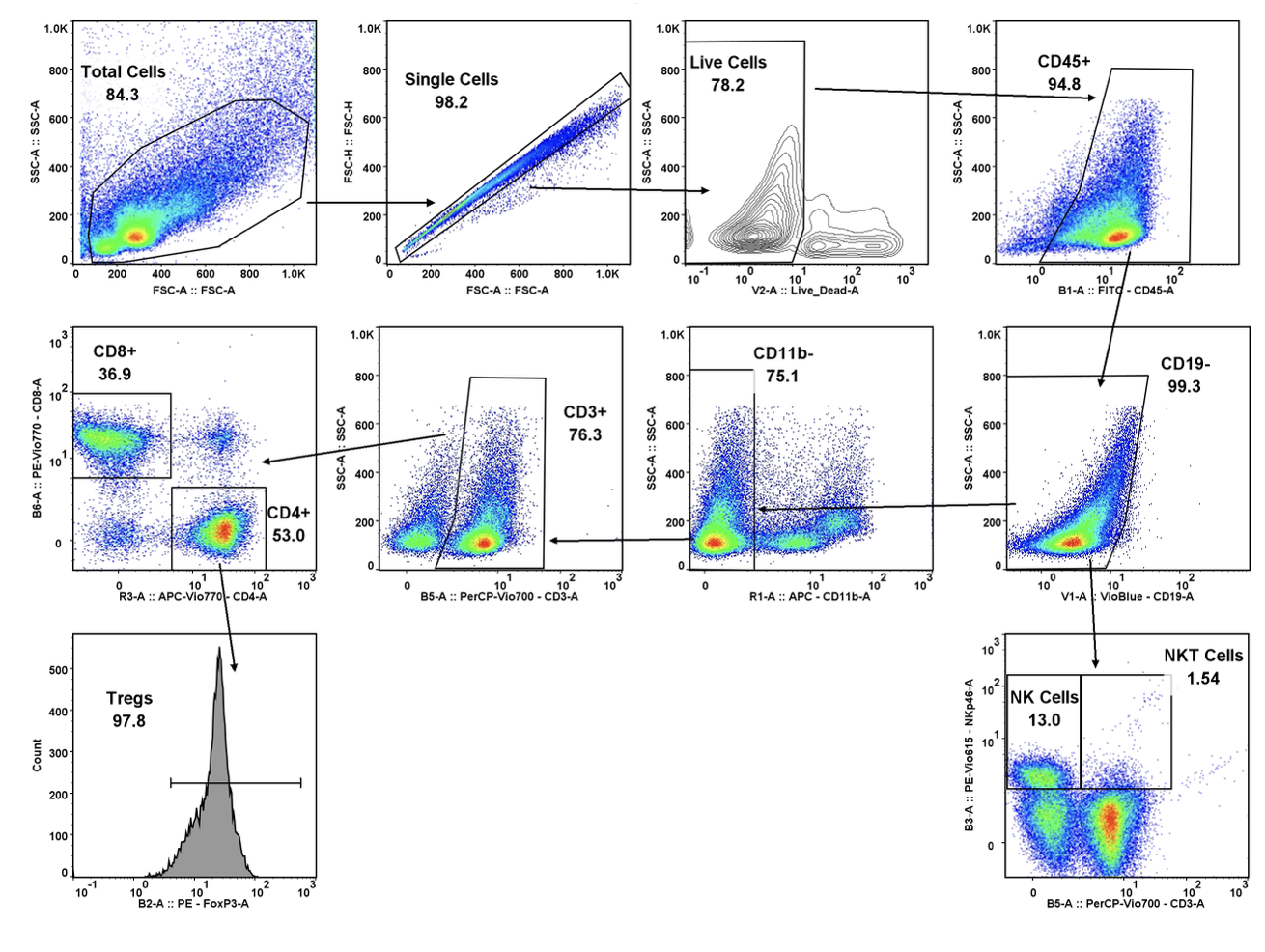
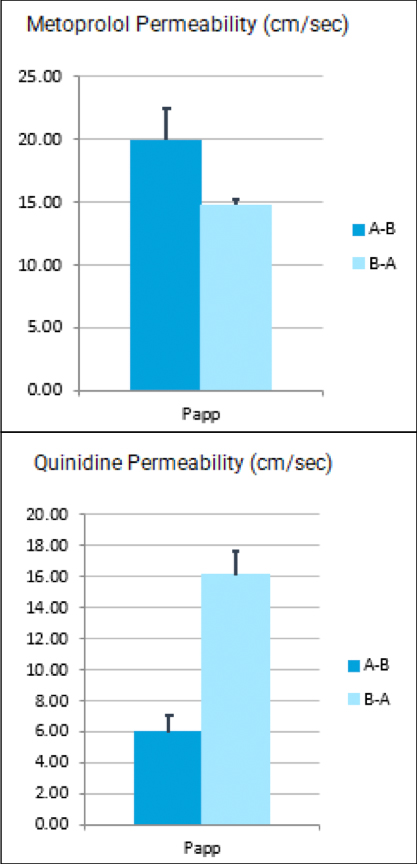
When it comes to oncology drugs, not all investigational drugs are available through an expanded access or special exemption mechanism. The main factors affecting an investigational drug’s availability outside of a clinical drug investigation include:
- The supply of the investigational drug available
- Patient demand for the investigational drug
- The degree to which the investigational drug sponsor determines that the product would have a notable impact on the patient’s current condition or prognosis
The most difficult part about participating in an expanded access program is getting access to the drug itself. Not all investigational drugs can provide the same benefits for the same patients, and a risk-benefit analysis has to be performed for each patient before an expanded access or special exemption mechanism can be deemed appropriate.
Additionally, an investigational oncology drug that’s too early on in the clinical drug investigation process would likely not have demonstrated a safe or even positive benefit.
Different pharmaceutical companies have different criteria for which investigational drugs can be used for which patients. The FDA is unable to compel a pharmaceutical company to offer such drugs to patients at all, and the agency has its own set of requirements.
Requirements for Access to Expanded Access Investigational Drugs
Under the Federal Food, Drug and Cosmetic Act (FD&C Act), a patient can pursue an investigational drug for the diagnosis, monitoring or treatment of cancer, provided that:
For more information about investigational new drugs, consider consulting the National Cancer Institute’s (NCI) PDQ database to track the status of ongoing studies. NCI’s Cancer Trials website, along with information specialists at NCI’s Cancer Information Service at 1-800-4-CANCER (1-800-422-6237), are also available to provide further information.
TD2 specializes in getting the best oncology treatments to patients who need them the most. Contact us for more information.
- Both the patient and doctor both want to participate in the process
- The physician agrees there is no equivalent therapy on the market to address the patient’s condition
- FDA determines that the investigational drug meets certain safety and efficacy requirements to treat the particular patient’s condition
- FDA determines that the possible risk to the patient posed by the investigational product is not higher than that of the cancer itself
- FDA determines that expanded access to the investigational drug will not in any way interfere with the ongoing clinical investigations and potential market approval
- The pharmaceutical company submits a clinical protocol for the patient that the FDA determines meets agency standards
- The patient is unable to participate in the associated clinical trial
For more information about investigational new drugs, consider consulting the National Cancer Institute’s (NCI) PDQ database to track the status of ongoing studies. NCI’s Cancer Trials website, along with information specialists at NCI’s Cancer Information Service at 1-800-4-CANCER (1-800-422-6237), are also available to provide further information.
TD2 specializes in getting the best oncology treatments to patients who need them the most. Contact us for more information.